Enterprise-grade post-market surveillance - built for teams of every size.
From complaint intake to adverse event reporting, recalls, risk management, and PMS reports, one connected workflow with AI embedded at every step. Whether you're a 20-person startup filing your first MDR or a global enterprise managing thousands, you get the same platform, the same AI, and the same compliance depth.
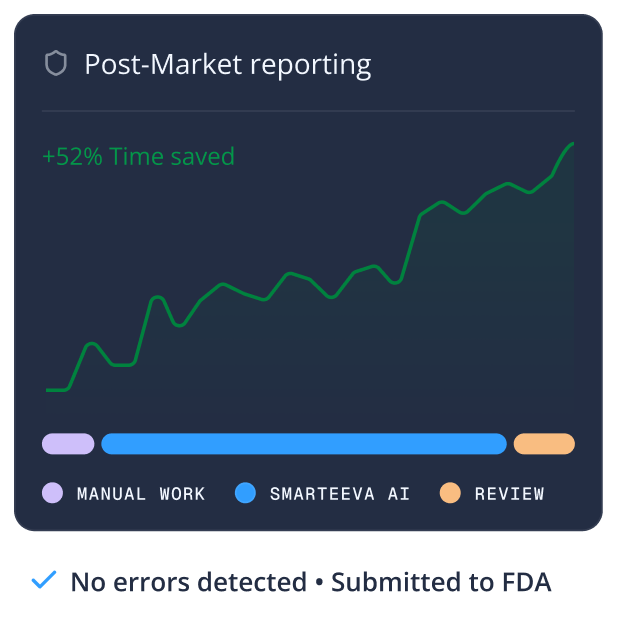




From first complaint to final regulatory submission - one platform for every step in between.



Built for the outcomes your leadership actually cares about.


The regulated industries that depend on us.
100% Salesforce-Native Platform
Smarteeva's AI continuously monitors complaint trends and adverse event patterns, flagging emerging safety signals before they become regulatory incidents or recalls.
What changes when your post-market workflow is purpose-built, not bolted on.
Other platforms
Built for document control across all industries. Post-market surveillance bolted on as an afterthought.
With Smarteeva
Built from day one for post-market surveillance. Every workflow, every AI model, every regulatory template designed for PMS.